Revolutionizing Lung Cancer Care with Cutting-Edge Genetic Testing
NGS Based Liquid Biopsy Test
What is LungTrack Advance?

LungTrack Advance is an NGS-based CAP-accredited liquid biopsy assay to screen all NCCN-guided actionable biomarkers. LungTrack Advance is performed on blood samples (Plasma cfDNA) to detect SNVs, Indels, and Fusions in all known/unknown fusion gene partners. It provides enhanced coverage of intronic regions for key fusion genes and their reported partners.

MedGenome Lung Cancer Liquid Biopsy Panel
- A next generation sequencing based CAP accredited assay to screen all the NCCN guided actionable biomarkers
- Only Blood is required; Test is performed on plasma cfDNA
- Detects SNVs, Indels and Fusions in 24 actionable genes
- Complete coding regions of the genes are covered; enhanced intronic region coverage for key fusion genes and their reported partners
- Provides results in 14 working days
Limit of Detection (LOD)
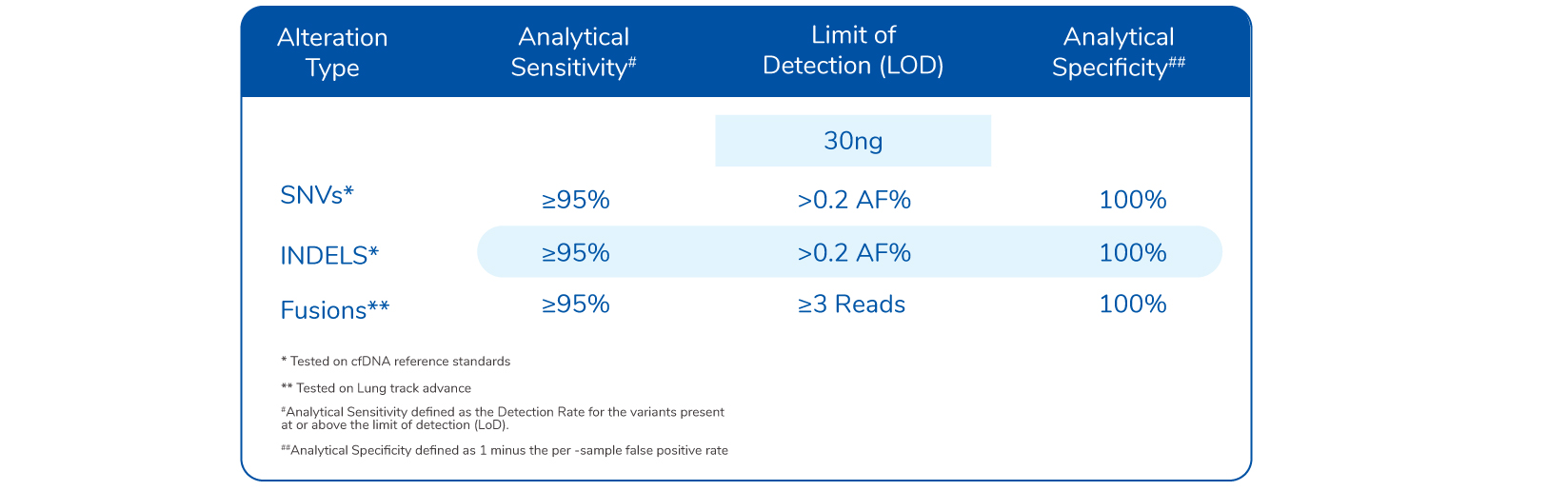
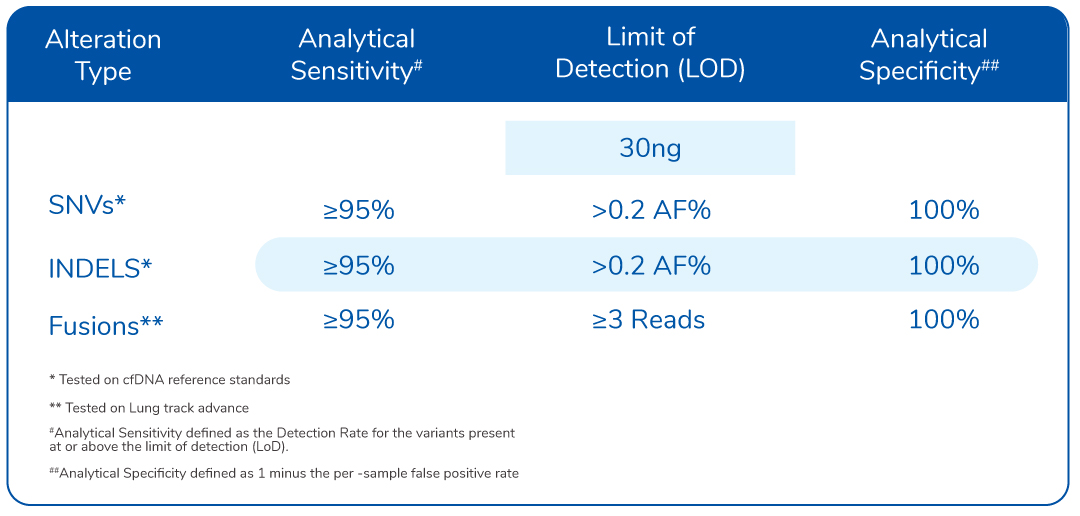
Assay specifications
| Cancer type | Specimen | Assay type | Sequencing Platform | Test Code | Average depth of sequencing |
|---|---|---|---|---|---|
| NSCLC | Peripheral Blood in Streck tube (10ml*2tubes) | UMI-based Next generation Sequencing | Illumina NGS platforms | MGM2623 | >20,000X (Pre UMI) >1000X (Post UMI) |
Gene List
| SNV’s and InDels | |||
| CTNNB1 | ERBB4 | PIK3CA | ERBB2 |
| ERBB3 | MAP2K1 (MEK) | TP53 | KIT |
| KRAS | CDKN2A | NRAS | EGFR |
| NTRK2 | NTRK3 | RET | ROS1 |
| ALK | FGFR2 | FGFR3 | NTRK1 |
| BRAF | NRG1 | MET | FGFR1 |
| Fusions | |||
| NTRK2 | NTRK3 | RET | ROS1 |
| ALK | FGFR2 | FGFR3 | NTRK1 |
| BRAF | NRG1 | MET | FGFR1 |
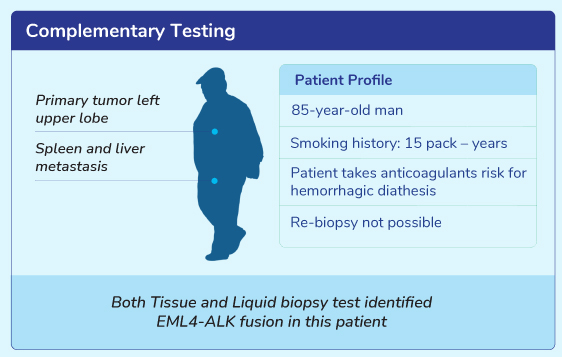
Clinical Scenarios For Liquid Biopsy Test

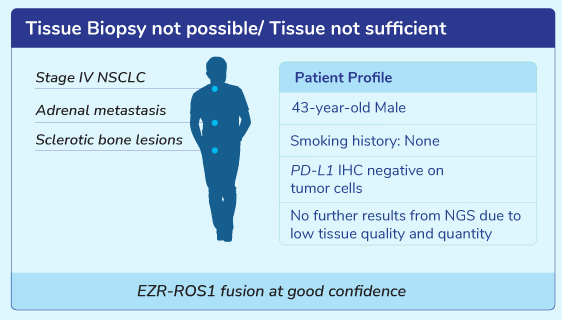
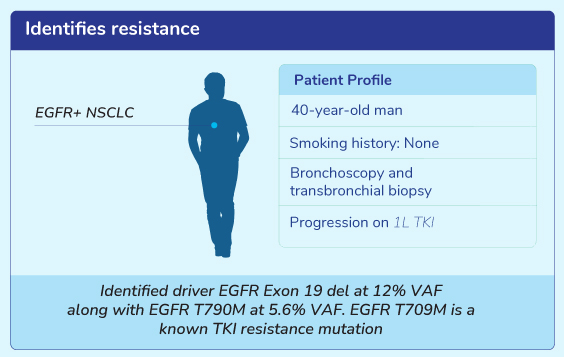
Get Genetic Counseling with MedGenome Genetic Experts
Please share your details with our genetic experts to answer your queries
